 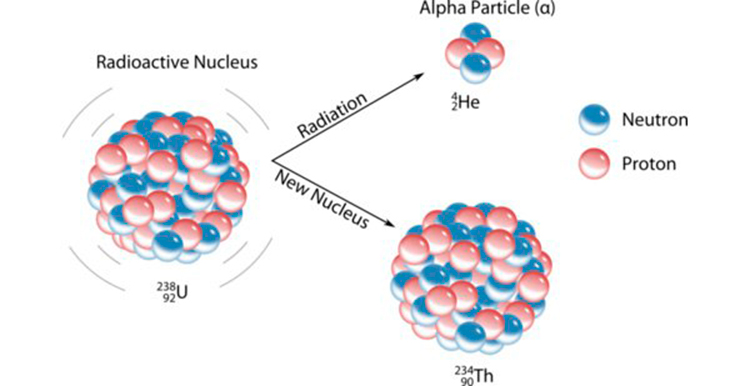
These example sentences are selected automatically from various online news sources to reflect current usage of the word 'alpha particle.' Views expressed in the examples do not represent the opinion of Merriam-Webster or its editors. 2018 Polonium-210 has a half-life of 139 days, meaning after this time half of its atoms have emitted an alpha particle and decayed into polonium-206 atoms. 2018 But unlike cosmic radiation or radon gas, plutonium can lodge itself inside the body and deliver tissue damaging alpha particles over a lifetime. 2018 But unlike cosmic radiation or radon gas, plutonium can lodge itself inside the body and deliver tissue-damaging alpha particles over a lifetime. 2012 These particles, primarily composed of electrons, protons, and helium nuclei (known as alpha particles), fly all over the solar system. Because 100 radioisotopes emit -particles, and most decay too quickly to be of therapeutic use, we will confine our discussion of -emitters to those that have. 2012 Like a bent arm, the Hoyle state takes the shape of an obtuse triangle with an alpha particle at each vertex. In this article, you will study alpha decay in detail. The deflection of alpha decay would be a positive charge as the particles have a +2e charge. Ernest Rutherford distinguished alpha decay from other forms of radiation by studying the deflection of the radiation through a magnetic field. 2019 Hoyle state nuclei almost always decay right back into beryllium and an alpha particle. This ejected particle is known as an alpha particle. 2020 Starting with oxygen gas, the team will split its nucleus into a helium nucleus, also known as an alpha particle, and a carbon-12 nucleus.ĭavid Grossman, Popular Mechanics, 21 Aug. Jacqueline Detwiler, Popular Mechanics, 1 Feb. Due to its short range,alpha particles emitted non-homogeneously from alpha particle sources on the skin surface will irradiate only a proportion of. If lead has sum of protons and neutrons 206, then it's mass should not be 206 u but 206.something uĪlso there is actual mass which we take by mass spectrometer.Recent Examples on the Web The fourth decay can occur by two different routes, with each path releasing one alpha particle and one beta particle before reaching lead-209. Why we take here lead mass as 206 u and alpha particle as 4u? So, definitely answer will be less then 5420 which is option Aĭoing it with hand fast 105/103 and then dividing 5420 will be difficult as you were stating, but we know that the answer will be less then 5420. Rutherford’s Alpha Particle Experiment Throughout the nineteenth century, scientists had the idea that each atom was composed of positive charges and these charges were distributed all over the atom. Yeah, we can do the conservation of momentum. What is Q? And how 5420 is 5407 in literature? Accuracy is pretty good: 1.2 keV in Po and Pb The 5407 is from here, they follow Wang et al. Alpha particles, also called alpha rays or alpha radiation, consist of two protons and two neutrons bound together into a particle identical to a helium-4. The results show a substantial improvement on earlier calculations. Bingo ! (But you knew the answer already ?) ALPHA-PARTICLE S-STATE BINDING ENERGY Full Record Related Research Abstract A variational calculation was made of the principal Sstate contribution to the binding energy of the alpha particle using a Yukawa interaction and a radial wave function. That division by 1.02 is the same as multiplying with 0.98, so subtracting 2%. In the last step you need to make a division, namely by approximately 1.02 (You still have to find out why). The 0.93 = 100% - 7% brings you to answer B (again, no coincidence). The mass of an alpha particle is therefore 4 amu, and its charge is +2. An alpha particle contains two protons and two neutrons, and no electrons. A and B differ 2%, so 1% should be enough already. An alpha particle is a free helium-4 nucleus. This means you can discard answer D and also C, but you still need some accuracy to distinguish between A and B. Next step is to go from μu to keV/c 2, the 0.9314941. Alpha radiation occurs when the nucleus of an atom becomes unstable (the ratio of neutrons to protons is too low) and alpha particles are emitted to restore. No multiplication, just a simple subtraction, to be done on paper with ease. (Which happens to be answer D - no coincidence). What you need to high accuracy is the difference (this value of Q). I wanted to postpone this story until you figured out the correct answer, but never mind: 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
